
Test Accuracy Ratio vs. Test Uncertainty Ratio

Consistent, accurate measurement results matter anywhere process decisions depend on equipment readings. In pharmaceutical manufacturing, this often includes pipettes and analytical balances. These are tools that directly affect batch composition and product quality.
One practical way to reduce measurement risk is to use an ISO/IEC 17025:2017 accredited calibration laboratory. Accreditation doesn’t always mean they are the best lab around. It only means the lab has been assessed against defined technical and quality requirements, including documented methods and uncertainty practices.
If you want to go one level deeper in vendor qualification, two common terms you may hear are Test Accuracy Ratio (TAR) and Test Uncertainty Ratio (TUR). They aren’t the only things that matter, but they can help you understand how a lab’s standards and methods compare to your equipment tolerances.
The Role of TAR and TUR in Pharmaceutical Equipment Calibration
To understand TAR and TUR, it helps to start with the basic calibration setup.
When a device is calibrated (often called the Unit Under Test, or UUT), the lab compares the UUT’s readings to a reference standard. The reference standard is the lab’s known measurement source (or the one they use to estimate the true value) used to evaluate the UUT.
Example: verification calibration of a PRT temperature probe
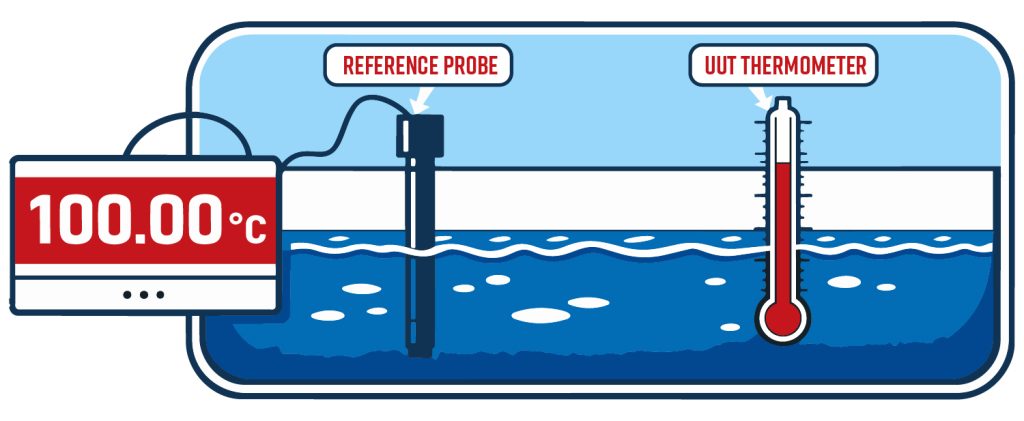
With that setup in mind, TAR and TUR are two ways people describe how good the reference/method is relative to the tolerances you care about.
What is the Test Accuracy Ratio (TAR)
Test Accuracy Ratio (TAR) compares the UUT’s stated accuracy to the reference standard’s stated accuracy.

Conceptually, TAR is trying to confirm that the reference standard is meaningfully more accurate than the thing being calibrated. And this is important because you want your calibration providers to use a reference standard that is more accurate than the device they are calibrating. This is not always possible, but it’s a very important concept, and if you are concerned about calibration quality, you can bring it up with your current and potential calibration providers.
What is the Test Uncertainty Ratio (TUR)
The Test Uncertainty Ratio (TUR) compares the UUT’s allowable tolerance to the calibration method’s combined uncertainty.
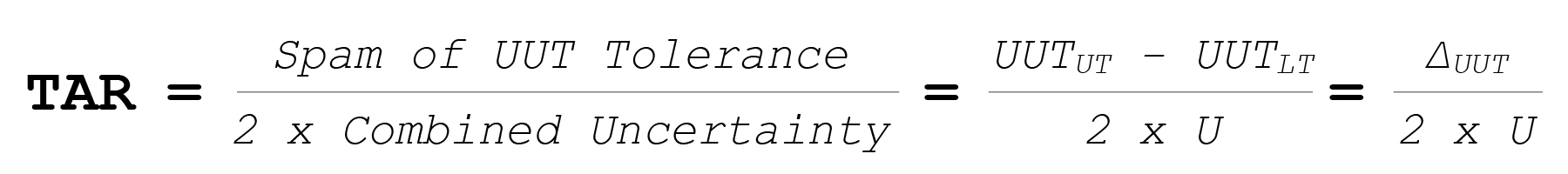
Where:

So:
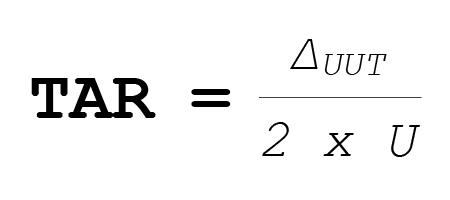
TUR matters because uncertainty is not just the reference standard accuracy. Uncertainty is a statistical estimate that accounts for multiple sources of variation and error in the calibration method. If you would like to learn more about the uncertainty of measurement, here is a link to one of our previous blogs: Blog 1 – What is Uncertainty Measurement?
Combined Uncertainty (U)
If the lab already has a calculated method uncertainty (often tied to the lab’s accredited capability), you can combine it with UUT-specific contributors, such as resolution and repeatability.
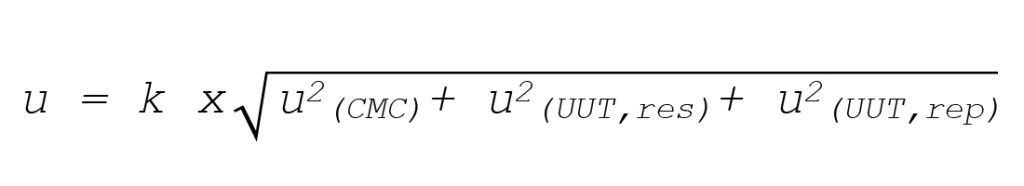
Where:

Should I Ask My Calibration Provider About TAR or TUR?
This is a great question, and many pharmaceutical and manufacturing companies don’t bother to ask their calibration providers about either one.
Many people consider TAR to be an older metric because it focuses on only two stated accuracies: the UUT and the reference standard. It does not explicitly include other contributors that can significantly affect real performance (e.g., setup, environment, operator technique, stability, resolution, etc.).
TUR is generally the most useful metric because it relates your tolerance to the method’s combined uncertainty. If you’re trying to evaluate risk, especially when you have tight tolerances, TUR gives a clearer picture of whether the calibration process can support meaningful pass/fail decisions.
If My Provider Is Not Providing TAR or TUR on the Certificate, Is That a Bad Thing?
No. Many labs do not print TAR or TUR on the calibration certificate, even when their methods and uncertainties meet industry standards and are reliable.
What would be concerning is if you ask the lab about TUR (or about uncertainty and how it’s determined), and they have difficulty providing an answer. A good calibration lab will have someone at the company who can answer questions about TAR, TUR, measurement uncertainty, and other technical details that may affect calibration results. If the lab is unable to answer your questions about the reference standard they use and how it compares to the UUT, you might want to reconsider having them perform the work. It is worth mentioning that sometimes good TAR and TUR values are not achievable for the work you are requesting. That doesn’t always mean the lab is not doing a good job. What you want in a laboratory can explain the risks involved in the calibration and provide you with options. You want a vendor that can clearly explain how their standards, methods, and uncertainties support the tolerances you have requested for your device’s calibration.
Conclusion
Using an ISO/IEC 17025:2017 accredited calibration laboratory is a practical way to reduce measurement risk, as accredited labs are assessed against defined technical and quality requirements. That matters in pharmaceutical environments where equipment results can affect product quality and compliance decisions.
TAR and TUR aren’t the whole story, but they’re useful checkpoints when you’re qualifying a provider, especially when your device has tight specifications, and you aren’t sure which devices could perform better than what you want calibrated. TUR is typically the more meaningful metric because it connects your tolerance to the calibration method’s combined uncertainty, not just stated accuracies.

If you would like to know more about selecting the right provider that will use appropriate standards when calibrating your equipment, please contact us at Bio Calibration Company. We would be happy to demonstrate how we strive to use the best equipment and standards available to calibrate our customers’ equipment and deliver the most reliable, repeatable calibration services.
